APOE*3-Leiden and APOE*3-Leiden.huCETP mouse model
Cardio Vascular and Metabolic disease models as Familial Hypercholesterolemia and Atherosclerosis
 APOE*3 Leiden transgenic mouse
APOE*3 Leiden transgenic mouse
This transgenic mice were generated by the introduction of the human apoe*3-Leiden and apoc1 genes.
The primary effect of the dominant E*3-Leiden mutation is an impaired clearance of triglyceride-rich lipoproteins (chylomicron- and VLDL-remnants) caused by reduced affinity for the LDLR, whereas overexpression of APOC1 inhibits lipolysis.
While normal wild-type mice have a very rapid clearance of apoB containing lipoproteins, E3L mice show an impaired clearance and are thereby mimicking the slow clearance in humans.
As a consequence, E3L mice exhibit a human-like lipoprotein profile comparable to that of patients with familial dysbetalipoproteinemia (most of the circulating cholesterol is contained to (V)LDL particles), and develop atherosclerosis upon feeding with saturated fat and cholesterol. However, E3L mice (like wild-type mice) do not possess a Cholesteryl Ester Transfer Protein (cetp) gene, an essential component of human lipoprotein metabolism, and therefore these mice do not respond to HDL-modulating interventions.
APOE*3-Leiden.huCETP (E3L.CETP) double transgenic mouse
This double transgenic mouse model has proven to be very suitable for testing the effects of drugs and bioactive nutritional components on plasma lipid levels, atherosclerosis, metabolic syndrome and MASH (NASH)-liver fibrosis. The model is based on TNO-Pharma’s proprietary mouse model, the Apolipoprotein E*3-Leiden (APOE*3-Leiden (E3L)) transgenic mouse, an established and well-recognized model for hyperlipidemia and the development of atherosclerosis (reviews Zadelaar et al. ATVB 2007; Kuhnast et al. Eur J Pharmacol 2015; Princen et al. Toxicol Rep 2016) and MASH (a.o. Morrison et al. J Hepatol 2015; van den Hoek et al. Hepatol Comm 2020; van den Hoek et al. Sci Rep 2021). In the E3L.CETP mouse human cholesterol ester transfer protein (huCETP) under control of its natural flanking regions was introduced into the E3L mouse resulting in a more human-like lipoprotein metabolism with transfer of cholesterol ester from HDL to the apoB-containing lipoproteins in exchange for triglycerides. As a result of this adverse lipoprotein distribution and the higher amount of atherogenic apoB-containing (non-HDL) lipoproteins, the E3L.CETP mice develop increased atherosclerosis on a Western-type diet as compared to E3L mice (Westerterp et al. ATVB 2006).
The E3L.CETP and E3L mouse models share the same favorable characteristics, such as (i) responsiveness to all hypolipidemic drugs currently used in the clinic, such as statins, fibrates, niacin, ezetimibe, and PCSK9 mAbs (Kuhnast et al. JLR 2014; Pouwer et al. JLR 2020) and ANGPTL3 mAbs (Dewey et al. NEJM 2017; Pouwer et al. JLR 2020) at similar dosages and in a similar way to humans, (ii) the ability to titrate cholesterol and triglycerides to any desired level and (iii) to conduct atherosclerosis studies in a progression (prevention) design or a regression (therapeutic) design (Pouwer et al. JLR 2020). Moreover, the APOE*3-Leiden.huCETP mouse is very well suited to testing the effect of drugs that modulate HDL and TG levels. The mice demonstrate reduced apoB-containing lipoproteins and increased HDL levels upon treatment with the registered drugs atorvastatin, fenofibrate and niacin, and with CETP-inhibitors (van den Hoek et al. DOM 2014; Kuhnast et al. Eur Heart J 2015).
In contrast to E-/- and LDLR-/- mice the E3L.(CETP) mice possess a functional apoE-LDLR-mediated clearance pathway for non-HDL lipoproteins, providing a suitable model for intervention studies with a.o. PCSK9 and ANGPTL3 inhibitors (Ason et al. JLR 2014; Dewey et al. NEJM 2017; Landlinger et al. Eur Heart J 2017; Suchowerska et al. JLR 2022; Zancanella et al. Mol Therapy Nucl Acids 2023). PCSK9 and ANGPTL3 inhibition with alirocumab or evinacumab demonstrated a significant additional effect on top of a statin with respect to lipid-lowering and atherosclerosis (Kuhnast et al. JLR 2014; Pouwer et al. JLR 2020), resulting in the first study in mice using this combination of clinical hypolipidemic drugs that shows true regression of atherosclerosis (Pouwer et al. JLR 2020).
APOE*3-Leiden/huCETP is a model for mixed dyslipidemia, a condition comparable with diabetic dyslipidemia and it is a predictive animal model: Drugs and nutritional components that failed in clinical studies, such as the GPR109a agonist MK-0354, the CETP-inhibitor torcetrapib (de Haan et al. Circulation 2008), an experimental RCT-inducer, plant sterol derivative and policosanols, also failed in this mouse model. With respect to biologicals, studies have been performed with mAbs, siRNA, miRNA, ASOs, peptides and vaccines against o.a. PCSK9, ANGPTL3, CETP, ox-LDL, Endothelial Lipase, IL-6, and with rec.HDL, GLP-1R, Y2-R and EPO-receptor agonists (a.o. Ason et al. JLR 2014; Kuhnast et al. JLR 2014; Landlinger et al. Eur Heart J 2017; Dewey et al. NEJM 2017; Zancanella et al. Mol Therapy Nucl Acids 2023, Inia et al. JLR 2025).
The mouse models have been used extensively in studies for pharma and food industry in testing efficacy and increasingly safety of small molecules and protein-based therapies and in studies towards the mechanism of action (> 200 studies).
Non-lipid targets and combination studies
The E3L and E3L.CETP mice are well-known mouse models for hyperlipidemia and atherosclerosis with a human-like lipoprotein metabolism which respond to all hypolipidemic drugs currently used in the clinic, such as statins, fibrates, niacin, ezetimibe and PCSK9 and ANGPTL3 inhibitors at similar dosages and in a similar way to humans. However, less well-known, the E3L and E3L.CETP mice are also suitable models for testing the efficacy of anti-inflammatory and blood pressure lowering drugs.
We have published a number of papers which show that modulation of non-lipid targets reduces progression of atherosclerosis, either alone or in combination with a statin, e.g. drugs/compounds with an anti-inflammatory/anti-oxidative action: resolvin E1 (Salic et al. Atherosclerosis 2016); epicatechin (Morrison et al. Atherosclerosis 2014); quercetin (Kleemann et al. Atherosclerosis 2011); salicylate (de Vries-van der Weij et al. Atherosclerosis 2010), all under pro-inflammatory conditions in E3L mice, in absence of cholesterol-lowering effects for resolving E1, epicatechin and quercetin.
And corticosterone (Auvinen et al. Plos One 2013), the SIRT1-activator resveratrol (Berbee et al. J Nutr Biochem 2013) and oncostatin M (van Keulen et al. Plos One 2019) under mild more human-like dietary conditions in E3L.CETP, without affecting plasma lipid levels for corticosterone and oncostatin M.
In addition, anti-hypertensive drugs with different modes of action such as the calcium antagonist amlodipine (Trion et al. J Cardiovasc Pharmacol 2006, efficacy independently of BP-lowering in E3L), the angiotensin II receptor blocker olmesartan (van der Hoorn et al. J Hypertension 2007, in E3L) and the renin inhibitor aliskiren (Kuhnast et al. J Hypertension 2012, in E3L.CETP) also reduce progression of atherosclerosis and improve plaque phenotype without effect on plasma cholesterol levels.
Moreover, the anti-inflammatory effects of a statin have been studied, in which the effects of rosuvastatin were compared to a low-cholesterol control group (matched to achieve the same plasma cholesterol levels of the statin-treated group) to allow investigation of the anti-inflammatory effects independent of the cholesterol-lowering effects of the statin (Kleemann et al. Circulation 2003).
New drugs need to show additional effect on top of a statin, considered to be the golden standard in the clinic. Since the E3L and E3L.(CETP) mice respond to statins as humans do, the model is very suited for combination studies.
Regression of atherosclerosis
With respect to plaque remodeling and regression of atherosclerosis, we recently showed for the first time in mice that true regression of pre-existent atherosclerosis, beyond the lesion size at the start of treatment, is possible in E3L.CETP mice with high-intensive cholesterol-lowering treatment using a combination of three drugs (Pouwer et al. JLR 2020: Alirocumab, evinacumab, and atorvastatin triple therapy regresses plaque lesions and improves lesion composition in mice). Detailed analysis of plaque composition in the latter study, but also from our other studies, indicate that next to regression of lesion size extensive remodeling takes place, with almost complete removal of macrophages and lipids from the plaques and increases in smooth muscle cells and collagen (e.g. Pouwer et al. JLR 2020; Landlinger et al. Eur Heart J 2017; Kuhnast et al. J Hypertension 2012; van der Hoorn et al. Brit J Pharmacol 2009).
Endothelial dysfunction
Together with the Jagiellonian Centre for Experimental Therapeutics at the Jagiellonian University, using sophisticated in vivo measurements of vascular function in the thoracic and abdominal aorta and flow-mediated dilatation in femoral artery using magnetic resonance imaging (MRI), we explored the effects of life-long mild hyperlipidemia on age-dependent endothelial dysfunction (Bar et al. Geroscience 2025). The study showed that endothelial function deteriorates with age, earlier in E3L.CETP male than in female mice. Further, E3L.CETP mice showed faster deterioration compared to wild-type mice, even before atherosclerosis develops. As one cannot study endothelial function and atherosclerosis separately in E-/- or LDLR-/- mice, the E3L.CETP mouse model provides the unique opportunity to study effects of compounds and drugs on endothelial dysfunction induced by dyslipidemia and ageing prior to atherosclerosis development. An example of is given in Kij et al. J Nutr Biochem 2025, showing for the first time that sufficient dietary phylloquinone (a member of the vitamin K family) intake supports endothelial function in normolipidemic and dyslipidemic E3L.CETP mice.
Metabolic syndrome model
In the E3L.CETP mouse on a high fat diet six important parameters of the metabolic syndrome are combined in one animal model which all change in the same direction as observed in humans, i.e. increased body weight and insulin resistance (increased plasma glucose and insulin levels) and at the same time adverse changes in plasma lipids as observed in diabetic dyslipidaemia, with increased triglycerides and apoB-containing lipoproteins and decreased HDL. This mouse model also develops MASLD (formerly known as NAFLD) . The model was validated by intervention with the anti-diabetic drugs rosiglitazone, the GLP-1R agonist liraglutide and an experimental 11β-HSD1-inhibitor, and the lipid-lowering drugs atorvastatin, fenofibrate and niacin, showing simultaneous decreases in plasma triglycerides, cholesterol, glucose and insulin with rosiglitazone, decreases in body weight and plasma glucose and insulin with liraglutide and the 11β-HSD1-inhibitor just as in humans, and decreases in plasma triglycerides and cholesterol with atorvastatin, fenofibrate and niacin, and increased HDL levels with fenofibrate and niacin, also just as in humans. In addition, hepatic triglycerides were significantly decreased by treatment with the high dose of rosiglitazone and liraglutide, while hepatic cholesterol esters were significantly decreased by the high dose of rosiglitazone and atorvastatin (van den Hoek et al. DOM 2014).
Mechanistic studies towards the etiology of high fat-induced insulin resistance and the contribution different organs showed metabolic dysregulation in liver, white adipose tissue and muscle with inflammatory responses in liver and WAT in response to accumulation of lipids (Kleemann et al. PLOS ONE 2010).
MASH-liver fibrosis
When fed a high fat/high cholesterol diet E3L and E3L.CETP mice develop metabolic dysfunction-associated steatohepatitis (MASH) (in about 12-16 weeks) and liver fibrosis (in 20-24 weeks). A general translational MASLD/MASH-liver fibrosis scoring system for rodent models was established based on characteristics in human MASH pathology (Liang et al. PLOS ONE 2014). The model has been validated with clinical candidates as obeticholic acid, elafibranor, icosabutate and several other drug and nutritional interventions (Morrison et al. J Hepatol 2015; Liang et al. Br J Pharmacol 2015; Zimmer et al. Hepatol Comm 2017; van den Hoek et al. Hepatol Comm 2020; van den Hoek et al. Sci Rep 2021; Inia et al. Int J Mol Sci 2023).
An unique feature using this model is the opportunity to conduct intervention studies with two or three relevant clinical endpoints in one animal study, atherosclerosis and MASH-liver fibrosis (Morrison et al. PLOS ONE 2015; Zimmer et al. Hepatol Comm 2017, Inia et al. Int J Mol Sci 2023, and a number of studies underway); and in combination with osteoarthritis (van Gemert et al. Osteoarthritis Cartilage 2021; van Gemert et al. Osteoarthritis Cartilage 2023 (2x)).
Cardiovascular safety
Moreover, due to its sensitivity for modulation of plasma lipids, parameters of the metabolic syndrome, inflammation and atherosclerosis adverse (side-) effects with respect to CV safety can be easily detected (e.g. of the CETP-inhibitor torcetrapib (de Haan et al Circulation 2008), the anti-tumor drug bexarotene (de Vries-van der Weij et al. Endocrinol 2009) and the drugs for chronic myeloid leukemia (CML) imatinib, nilotinib and ponatinib (Pouwer et al. Front Cardiovasc Med 2018; Carracedo et al. Brit J Pharmacol 2022), mineral oil (Pieterman et al. Front Pharmacol 2021), HIV-protease and reverse transcriptase inhibitors, drugs against Gaucher’s Disease, 11β-HSD1-inhibitor, JAK-inhibitors, plant sterol derivative).
With respect to environmental safety and related CV safety issues a series of studies towards the mechanism of action of perfluorosurfactants (resistant to (bio)degradation) on lipid and lipoprotein metabolism were conducted (Bijland et al. Toxicol Sci 2011; Pouwer et al. Toxicol Sci 2019).
Track record
The TNO group has a strong academic background as indicated by the publication list containing many papers on the mechanism of action of drugs, pleiotropic and side effects, often conducted together with partners from industry.
During the past 20 years the department has conducted over 250 projects for industrial partners from pharmaceutical, food and nutrition industry. During the past 3 years on average 15-20 industrial projects per year have been conducted. Client satisfaction score is on average 4.7-4.8 on a scale of 5.
Download
□ Models for Cardiovasucular and Metabolic diseases
□ Cardiovasucular Safety
□ Publication list. ver.Jan-2026
□
Proof-of-concept study for liver-directed miQURE technology in a dyslipidemic mouse model. Molecular Therapy Nucleic Acids, June 2023
Latest publication
□ Efficacy of a novel PCSK9 inhibitory peptide alone and with evinacumab in a mouse model of atherosclerosis.
J Lipid Res 2025; Inia JA, van Nieuwkoop-van Straalen A, Jukema JW, Rolin B, Staarup EM, Mogensen CK, Princen HMG, van den Hoek AM.
□ Obicetrapib and ezetimibe enhance LDL receptor-mediated VLDL-clearance and regress atherosclerosis on atorvastatin background.
J Lipid Res 2026 (conditionally accepted); Inia JA, Zhang L, Keijzer N, Worms N, van Nieuwkoop-van Straalen A, Ditmarsch M, de Kleer M, Jukema JW, Kastelein JJP, Szarek M, van den Hoek AM, Stokman G, Pieterman EJ, Princen HMG.
AI-Powered Preclinical Atherosclerosis Research
The APOE3-Leiden (E3L) and APOE3-Leiden.huCETP (E3L.CETP) mouse models are well-established, translational models for evaluating therapies targeting plasma lipids, lipoprotein metabolism, and atherosclerosis, with proven responsiveness to clinically used lipid-lowering drugs in both preventive and therapeutic study designs. AI-assisted assessment enhances these studies by standardizing lesion quantification, reducing inter- and intra-reader variability, and improving sensitivity to detect treatment effects. TNO Athero AI transforms routine H&E-stained aortic root sections into objective, quantitative datasets that strengthen confidence in your efficacy data.
 Score with consistency
Score with consistency
Increase scoring confidence by minimizing variability in atherosclerosis trials and research. TNO Athero AI ensures that every lesion is evaluated against the same objective criteria, eliminating the subjective differences that arise between human readers and across different reading sessions.
Segment and classify
TNO Athero AI accurately segments plaques and characterizes their composition, including necrotic core, calcification, fibrous tissue, and smooth muscle content. This detailed compositional analysis reveals treatment effects that conventional area-based measurements would miss entirely.
Efficient workflows
Speed up trials by reducing turnaround times through AI-assisted reading. Automated analysis processes hundreds of whole-slide images in a fraction of the time required for manual pathologist review, enabling faster data delivery without compromising quality or detail.
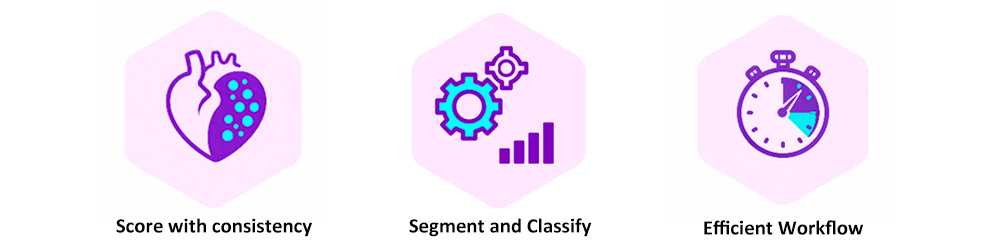
Precise and standardized plaque readout
TNO Athero AI segmentation enables precise and standardized plaque readout, accurately delineating lesion boundaries across the complete aortic root cross-section. Automated analysis reduces turnaround time and minimizes reader variability, while clear, color-coded outputs make results straightforward to interpret in atherosclerosis research and preclinical efficacy studies. Each plaque component is mapped with pixel-level precision, providing quantitative measurements that go far beyond what manual assessment can achieve. AI segmentation accurately delineates lesion boundaries across the complete aortic root cross-section, mapping each plaque component with pixel-level precision. Small, early-stage lesions are consistently detected.
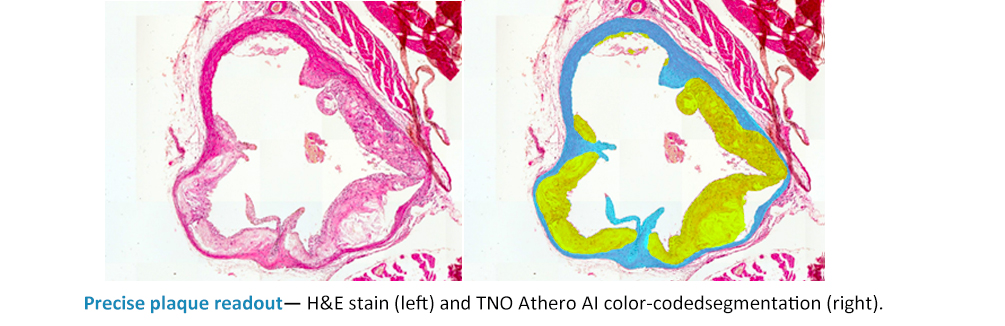
Consistent detection of early-stage lesions
TNO Athero AI analysis helps ensure that even small, early-stage atherosclerotic lesions are consistently detected and accurately delineated across all study samples. In preventive study designs where lesion burden may be significantly reduced by effective treatment, this sensitivity is critical. The AI model reduces the risk of overlooked features and strengthens the reliability of lesion assessment within and across studies, ensuring that subtle but meaningful treatment effects are captured with statistical confidence.
How HistoSuite Works
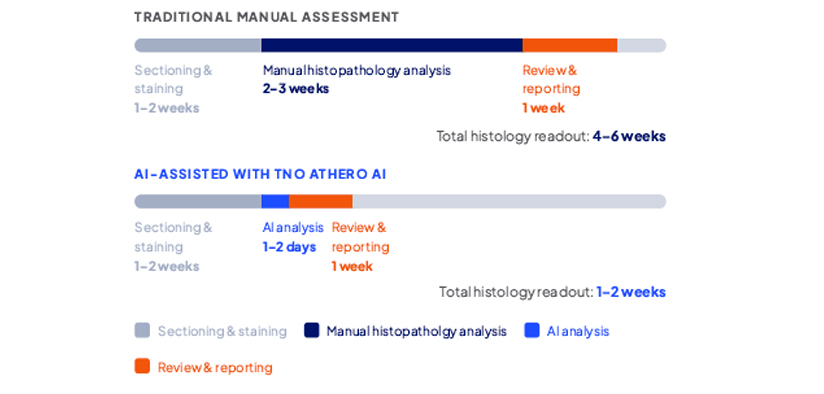 By automating the most labor-intensive step in plaque assessment, TNOAthero AI compresses analysis windows from weeks to days ? without sacrificing depth, accuracy, or reproducibility.
By automating the most labor-intensive step in plaque assessment, TNOAthero AI compresses analysis windows from weeks to days ? without sacrificing depth, accuracy, or reproducibility.

