in vitro and in vivo models to study mechanisms, interventions and biomarkers
in metabolic diseases and their complications.
Metabolic Health Research (MHR) at TNO
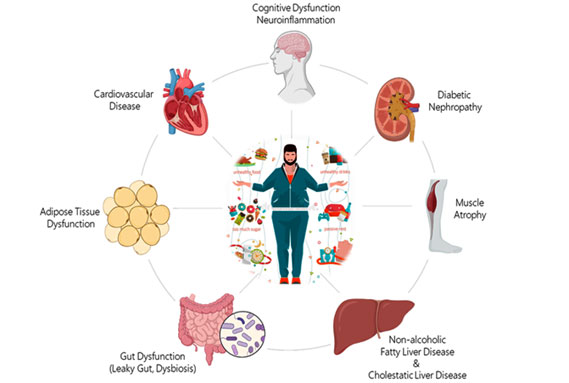
MHR develops and performs in vitro and in vivo models to study mechanisms, interventions and biomarkers in metabolic diseases and their complications.
These translational models include unique (humanized) transgenic mouse models, in vivo and in vitro models, read-out systems employing i.a. histology, biochemical assays, cell biology, molecular biology, immunology and inflammation markers.
This preclinical research is strongly translational and aims to improve the predictability of efficacy and safety of pharmaceutical and food interventions by detailed knowledge of disease processes and mechanisms.
MHR has a track record in applied science, study design, professional project management and quality systems.
MHR offers customized services that can be tailored to customer needs by direct interaction of scientists of MHR and the customer.
We also propose partnering opportunities to gain a deeper understanding of the following points:
● Elucidation of disease mechanisms at the molecular level
● Elucidation of organ cross talk during the disease onset process
● Identification of novel biomarkers
Disease area of Interest
● Obesity, MASLD / MASH
● Sarcopenia / Frailty
● Cardiovascular-Kidney-Metabolic (CKM) syndrome・Heart Failure(HFpEF)
● Woman's Health (Endometoriosis, Menopause, etc)
Women's Health;Health and lifestyle

Our understanding of the female body and its unique healthcare needs remains surprisingly limited. Many drugs are mainly tested on men, and it was only recently that female-specific heart disease symptoms were recognized. Conditions like endometriosis often take an average of 10 years to diagnose. This health disparity also impacts labour productivity, costing billions globally.
TNO's Women's Health program integrates our biomedical, (psycho)social, and technological expertise to tackle women's health challenges. By merging fundamental research with practical solutions, we aim to ensure that future generations of women can achieve their full potential without health-related barriers. However, this goal cannot be reached in isolation.
We collaborate with healthcare professionals, employers, researchers, policymakers, and companies, including those in the food, pharmaceutical and technology sectors.
Current co-development & collaboration opportunities
● Endometriosis: early warning system biomarkers, microbiome and efficacy testing.
● Muscle aging and frailty: cardiac disease, frailty and sarcopenia, optimal performance and sustained employability throughout life.
● Menopause and sex hormones: Key disease drivers and efficacy testing on fibrosis.
● Vaginal health: new in-vitro screenings model.
● Women, work and family health: scan for employers, life cycle perspective on employability, systemic interventions (community approaches, chain approaches), eHealth technologies.
Cardiovascular-Kidney-Metabolic (CKM) syndrome model
 Cardiovascular-Kidney-Metabolic (CKM) Syndrome is a complex disorder involving CVD, kidney disease, type 2 diabetes and obesity.
These conditions share risk factors and can exacerbate each other. Development of new therapies is hampered by the lack of translational models.
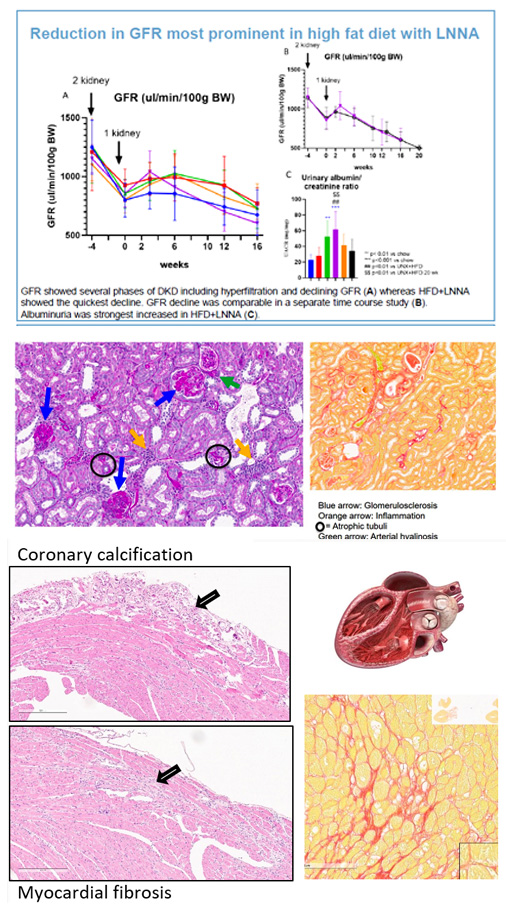
A novel diet induced hypertension-accelerated mouse model with obesity, diabetes and hypertension was developed which shows diabetic and chronic kidney disease (DKD/CKD) and heart failure with preserved ejection fraction (HFpEF).
Male KKAy mice on HFD and LNNA developed DKD resulting in CKD and HFpEF.
Combination therapy with Lisinopril and Dapagliflozin rescued GFR decline and reduced renal and cardiac damage.
This indicates the clinical relevance of the model allowing holistic and mechanistic studies in both early and more advanced stages of CKM Syndrome.
Cardiovascular-Kidney-Metabolic (CKM) Syndrome is a complex disorder involving CVD, kidney disease, type 2 diabetes and obesity.
These conditions share risk factors and can exacerbate each other. Development of new therapies is hampered by the lack of translational models.
A novel diet induced hypertension-accelerated mouse model with obesity, diabetes and hypertension was developed which shows diabetic and chronic kidney disease (DKD/CKD) and heart failure with preserved ejection fraction (HFpEF).
Male KKAy mice on HFD and LNNA developed DKD resulting in CKD and HFpEF.
Combination therapy with Lisinopril and Dapagliflozin rescued GFR decline and reduced renal and cardiac damage.
This indicates the clinical relevance of the model allowing holistic and mechanistic studies in both early and more advanced stages of CKM Syndrome.
Features for Cardiovascular-Kidney-Metabolic (CKM) syndrome model
・Progressive decline in renal function in setting of hyperglycemia,
typically preceded by a period of glomerular hyperfiltration.
・Quick onset of Albuminuria
・Pathological changes in kidneys:
> Glomerular basement membrane thickening
> Mesangial matrix expansion and sclerosis
> Tubulo-interstitial fibrosis
> Arteriolar hyalinosis
・Diet induced (Human-like diet composition)
・The model shows hyperlipidemia, hyperglycemia and hypertension, albuminuria, decline in GFR and typical histological features of DKD.
・The model also shows Coronary calcification, Myocardial fibrosis, HFpEF and HFrEF as observed in Heart Failure.
・Reference control: Combination of ACE inhibitor + SGLT2 ihibitor
Readout parameters
・Metabolic parameter: BW, F&W-intake, Glucose, Insulin, CHO, TG
・Function: Diuresis, Albuminuria, UACR, GFR by TD inulin clearance
・Pathology: Quantitative scoring of Glomerular and Tubular damage, Liver and Cardiac damage
・Optional: Proteomics/Metabolomics,Next generation sequencing, EM microscopy, PEMP analysis (NIPOKA)
Download Publication
□
Treatment with Either SGLT2 Inhibitors or GLP-1 Receptor Agonists Rescues GFR Decline in a Multifactorial Cardiovascular-Kidney-Metabolic Syndrome Mouse Model.Journal of the American Society of Nephrology
□
Standard-of-care combination therapy rescues GFR decline and cardiac damage in a novel cardiovascular-kidney-metabolic syndrome mouse model
□
Combination therapy with Lisinopril and Dapagliflozin rescues GFR decline and glomerular damage in the advanced DKD/CKD KKAY mouse model.
□
Cardiac damage in DKD/CKD mouse model resembles HFpEF and can be reduced by Standard-of-care treatment.
Sarcopenia, Frailty models
 The prevalence of sarcopenia is increasing and effective interventions are required to prevent or reverse age-related muscle loss. However, it often is challenging, expensive and time-consuming to develop and test the effectiveness of such interventions and translational animal models that are adequately mimicking the underlying physiological pathways are scarce. Strong predictors for the incidence of sarcopenia include a sedentary life-style and malnutrition.
TNO has therefore recently developed a new and short (2 weeks) mouse model for muscle atrophy that combines caloric restriction with partial immobilization (of one hindleg). This combination model exhibits loss of muscle mass and function. Transcriptome analysis demonstrated that the underlying pathways of this combination model revealed more similarity with the human underlying pathways than aged mice.
In addition to the combination models, we previously demonstrated a beneficial treatment effect in the CR alone model (van den Hoek AM et al., Metabolism 2019).
In addition to the abovementioned models, TNO has several technologies (including AMS; Low dose 14C-alanine incorporation to enable high sensitive detection and tracing of protein turnover in muscle) available for muscle related readouts. In addition, we perform biomarkers research and can support companies with sensors/eHealth solutions in this therapeutic indication.
The prevalence of sarcopenia is increasing and effective interventions are required to prevent or reverse age-related muscle loss. However, it often is challenging, expensive and time-consuming to develop and test the effectiveness of such interventions and translational animal models that are adequately mimicking the underlying physiological pathways are scarce. Strong predictors for the incidence of sarcopenia include a sedentary life-style and malnutrition.
TNO has therefore recently developed a new and short (2 weeks) mouse model for muscle atrophy that combines caloric restriction with partial immobilization (of one hindleg). This combination model exhibits loss of muscle mass and function. Transcriptome analysis demonstrated that the underlying pathways of this combination model revealed more similarity with the human underlying pathways than aged mice.
In addition to the combination models, we previously demonstrated a beneficial treatment effect in the CR alone model (van den Hoek AM et al., Metabolism 2019).
In addition to the abovementioned models, TNO has several technologies (including AMS; Low dose 14C-alanine incorporation to enable high sensitive detection and tracing of protein turnover in muscle) available for muscle related readouts. In addition, we perform biomarkers research and can support companies with sensors/eHealth solutions in this therapeutic indication.
Model option in vitro; in vitro muscle model using C2C12 cells
Readout parameters
・Cell proliferation (number of nuclei per field)
・Differentiation (number of myotubes per field, fusion index, area and diameter of myotubes)
Available technology; EPS (Electrical Pulse Stimulation) to induce contraction
□ Publication:
In vitro muscle contraction: A technical review on electrical pulse stimulation in C2C12 cells, Exp Physiol, Aug 2025
(Watch the video; C2C12 cells that contract 1 second, followed by 4 seconds of rest)
Model options in vivo; C57BL6 mice Regimen; either prophylactic, or therapeutic.
1: Caloric restriction model.
2: Combination model: combining caloric restriction and immobilization.
3: Aged mice.
Readout parameters
・Echo-MRI: Lean body mass/body composition
・Functional tests: Grip strength, inverted screen, voluntary movement/physical activity
・Histology: Cell diameter; slow/fast fiber type, collagen
・Transcriptomics: Pathway analysis, super regulator prediction
・Protein signalling pathways: Pathway analysis (insulin, mTOR, AMPK, FOXO)
・Biochemical: Intramuscular triglyceride content
・AMS: Low dose 14C-alanine incorporation to enable high sensitive detection and tracing of protein turnover in muscle
・Deuterated water can be used for measurement of protein synthesis rate.
Recent Publication:
□ Anabolic effects of salbutamol are lost upon immobilization. J Cachexia Sarcopenia Muscle 2025; 16: e70114. doi.org/10.1002/jcsm.70114.
de Jong JCBC, Jameson TSO, Andrews RC, Dunlop MV, Abdelrahman DR, Murton AJ, Caspers MPM, Worms N, van Nieuwkoop A, Keijzer N, Cheng Q, GuigasB, van Duijn E, Vaes WHJ, Nieuwenhuizen AG, Keijer J, Wall BT, Verschuren L, Stephens FB,| van den Hoek AM, Dirks ML.
□ Blood-based biomarkers for early frailty are sex-specific: validation of a combined in silico prediction and data-driven approach. Geroscience 2025; 47:3741-3758. doi: 10.1007/s11357-024-01449-w.
de Jong JCBC, Caspers MPM, Dulos R, Snabel J, van der Hoek MD, van der Leij FR, Kleemann R, Keijer J, Nieuwenhuizen AG, van den Hoek AM, Verschuren L.
van de Meene MRC, van den Hoek AM, Hanemaaijer R, Verschuren L, de Jong JCBC. In vitro muscle contraction: A technical review on electrical pulse stimulation in C2C12 cells. Exp Physiol 2025; 1-16. DOI: 10.1113/EP092677.
□ Translatability of mouse muscle-aging for humans: the role of sex. Geroscience 2024; 46: 3341-3360. doi: 10.1007/s11357-024-01082-7.
de Jong JCBC, Caspers MPM, Worms N, Keijzer N, Kleemann R, Menke AL, Nieuwenhuizen AG, Keijer J, Verschuren L, van den Hoek AM.
□ Caloric restriction combined with immobilization as translational model for sarcopenia expressing key-pathways of human pathology. Aging Dis 2023; 14: 937-957. doi: 10.14336/AD.2022.1201.
de Jong JCBC, Caspers MPM, Keijzer N, Worms N, Attema J, de Ruiter C, Lek S, Nieuwenhuizen AG, Keijer J, Menke AL, Kleemann R, Verschuren L, van den Hoek AM.
□ Diet and exercise reduce pre-existing NASH and fibrosis and have additional beneficial effects on the vasculature, adipose tissue and skeletal muscle via organ-crosstalk. Metabolism 2021;124:154873. doi: 10.1016/j.metabol.2021.154873.
van den Hoek AM, de Jong JCBC, Worms N, van Nieuwkoop A, Voskuilen M, Menke AL, Lek S, Caspers MPM, Verschuren L, Kleemann R.
□ Intramuscular short-chain acylcarnitines in elderly people are decreased in (pre-)frail females, but not in males. FASEB J 2020; 34:11658-11671. Doi: 10.1096/fj.202000493R.
van der Hoek MD, Nieuwenhuizen AG, Kuda O, Bos P, Paluchova V, Verschuren L, van den Hoek AM, Kleemann R, Veeger NJGM, van der Leij FR, Keijer J.
□ A novel nutritional supplement prevents muscle loss and accelerates muscle mass recovery in caloric-restricted mice. Metabolism 2019;97:57-67. doi: 10.1016/j.metabol.2019.05.012.
van den Hoek AM, Zondag GCM, Verschuren L, de Ruiter C, Attema J, de Wit EC, Schwerk AMK, Guigas B, Lek S, Rietman A, Strijker R, Kleemann R.
Collaboration program
In addition, TNO is leader of a research consortium that focuses on a better understanding of sarcopenia, including the underlying mechanism.
An unique human cohort is studied to further validate the translatability of our models and to identify biomarkers.
In addition, male/female differences will be studied.
Future directions
・We are also investigating Muscle Health in relation to cognition.
・Muscle health in relation to menopause.
・Muscle Health in relation to the use of GLP-1 analogues (semaglutide/Ozempic).
Fibrosis models
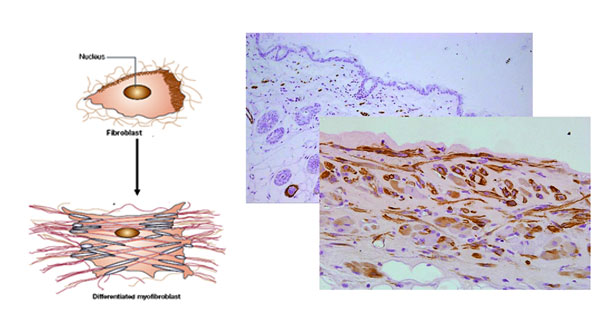
・Lung Bleomycin-induced lung fibrosis model in mice
(Feature:o.p.administration, low variation and motality)
・Skin Bleomycin-induced skin fibrosis model in mice.
・Liver CCL4-induced, Diet-induced model in mice
・Kidney UUO model in mice.
・in vitro fibrosis assay with the fibrosis patient samples
Myoblast differenciation, Fibroblast proliferation, Migration
Current co-development & collaboration opportunities
● Development of Novel Blood-Based Biomarkers for IPF
Recent publication: Targeting the Wnt signaling pathway through R-spondin 3 identifies an anti-fibrosis treatment strategy for multiple organs
We're open to discuss;
・Collagen type analysis:Collagen 1α1, 3α1, 4α1, 5α1, 6α2.
・Signatutre analysis involving newly synthetised collagen
Reference
□ Collagen quantification in cell cultures and tissues.

